Research in the Masri lab is aimed at understanding the relationship between disruption of circadian rhythms and tumor initiation and progression. The circadian clock sustains self-perpetuating oscillations with a 24-hour periodicity. These circadian rhythms include sleep-wake cycles, feeding behavior, endocrine oscillations, metabolic control, and immunity (Fortin et al., Nature Cancer, 2025; Verlande et al., Trends in Endocrinology and Metabolism, 2019). Alterations in biological rhythms result in numerous physiological disorders, the consequences of which have been linked to several pathologies, including cancer. The molecular machinery that constitutes the circadian clock is comprised of two transcription factors, CLOCK and BMAL1, that heterodimerize and direct transcriptional activation of core clock genes and additional clock-controlled genes (CCGs), as shown below.
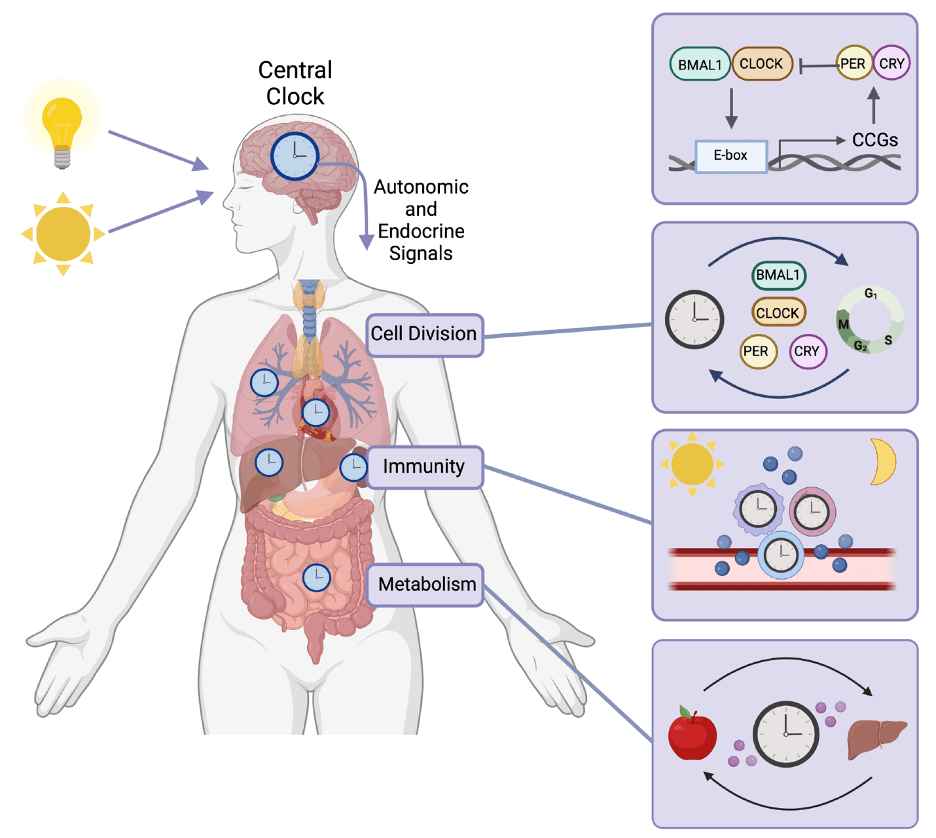
The circadian clock, oncogenic signaling, and genome instability
Adenomatous polyposis coli (APC) mutations have been reported in 80% of human colorectal cancer cases and these mutations drive initiation of intestinal adenoma development. The Masri lab has reported that genetic and environmental disruption of the circadian clock accelerates colorectal cancer progression in vivo (Chun, Fortin, Fellows et al., Science Advances, 2022). Using intestinal organoids, we demonstrate that clock disruption promotes transformation by driving Apc loss of heterozygosity (LOH), which hyperactivates Wnt signaling to drive colorectal cancer. The Masri lab is currently exploring how perturbation of circadian rhythms impacts genome instability and the molecular underpinnings driving Apc LOH. Our findings demonstrate a new mechanistic link between clock disruption and colorectal cancer, which has important implications for cancer prevention and therapeutic targeting.
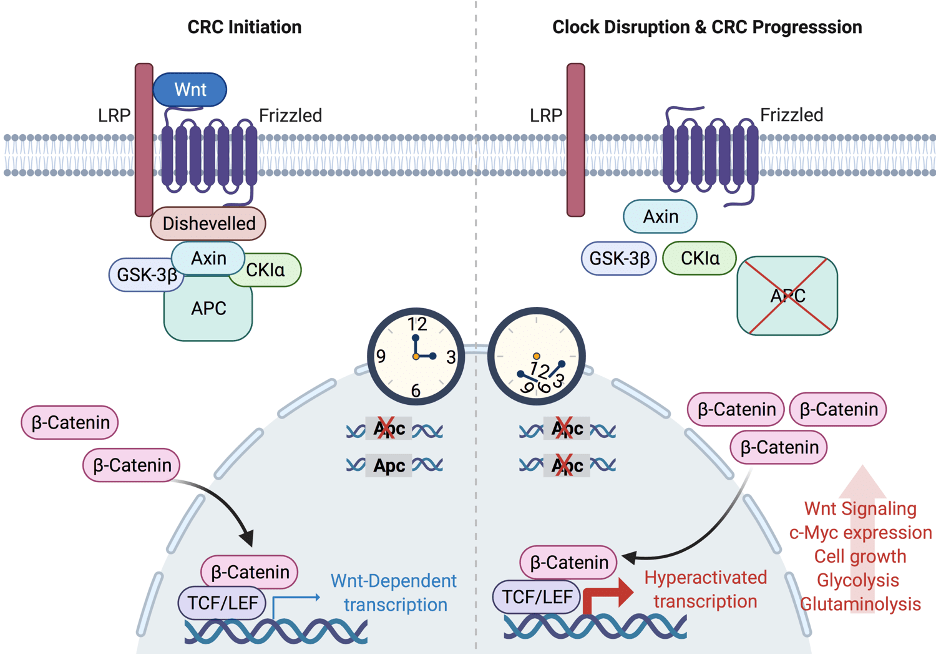
The circadian clock and anti-tumor immunity
The circadian clock is known to impinge on the immune landscape to regulate the abundance and activity of myeloid and lymphoid cells involved in innate immunity. However, less is known regarding the time-of-day dependent control of anti-tumor immunity and how alterations in circadian rhythms could impact immunosurveillance. Using genetically engineered mouse models (GEMMs) of lung and colorectal cancer, the Masri lab is utilizing unbiased single cell RNA-sequencing and downstream applications to interrogate how the circadian clock governs anti-tumor immunity. Additionally, using this knowledge, the Masri lab is leveraging these findings to improve therapeutic potential of immunotherapy based on time-of-day delivery, an approach called chronomedicine (Fortin et al., Nature Immunology, 2024).

The circadian clock, nutrition, and stemness in cancer
Intestinal stemness is tightly governed by key signaling pathways, and aberrant activation of these pathways plays an important role in colorectal cancer. The Masri lab is dissecting how the circadian clock impacts intestinal stemness that is implicated in colorectal cancer. Also, the role of nutritional cues, that can impinge on the circadian clock and in turn regulate these signaling networks, are being dissected. This work is being carried out using mouse models in vivo, as well as ex vivo using mouse intestinal organoids that model mini-gut structures in a 3D matrix (see pictures below). These studies have very important clinical implications in understanding how disruption of the biological pacemaker, on the molecular level, alters tumor initiation and disease progression.

The circadian clock and systemic cancer metabolism
The Masri lab is elucidating the systemic crosstalk between tumors and peripheral tissues in the context of cancer cachexia. Cachexia-associated cachexia is a wasting syndrome associated with loss of adipose tissue and skeletal muscle that results in systemic metabolic alterations. Our previous studies have shown that lung adenocarcinoma and development of cachexia are associated with rewiring of circadian metabolism in the liver (Masri et al., Cell 2016) through a key clock factor, Rev-ERBa (Verlande et al., Science Advances, 2021). We are characterizing the biological consequences of tumor-dependent metabolic intermediates in circulation to define if tumors can repurpose metabolic intermediates for energy production. To address these questions, we utilize metabolomics and stable isotope-based approaches in vivo. Importantly, we aim to identify key tumor-dependent metabolic intermediates and the host cellular pathways responsible for metabolic repurposing, as a means to establish novel therapeutic modalities for starving tumors.


